The conformation of the hydroxymethyl group in carbohydrates is best described
as an equilibrium between three staggered rotamers. In hexopyranoses and pentofuranoses
they are referred to as gt (gauche-trans), gg (gauche-gauche)
and tg (trans-gauche).
The corresponding torsion angles between the terminal oxygen
and the ring oxygen (ω) are +65°, -65° and 180°.
Hydroxymethyl rotamers in hexopyranoses
|
|

|
| gauche-trans rotamer |
gauche-gauche rotamer |
trans-gauche rotamer |
| ω=+65° |
ω=-65° |
ω=180° |
| No unfavourable 1,3-interactions |
Unfavourable 1,3-interactions in galacto-compounds |
Unfavourable 1,3-interactions in gluco-compounds |
| Favoured by the gauche-effect |
Favoured by the gauche-effect |
|
The common method for determining the relative rotamer populations is to use the proton-proton
couplings between H5 and H6proR and H6proS in hexoses
(or H4 and H5proR and H5proS in pentoses).
This requires the resonances of the prochiral hydroxymethyl protons to be assigned.
In general the HproR resonance will appear upfield of the HproS resonance. In derivatives that are acetylated
or α-D-glycosylated
at the hydroxymethyl group the order of the resonances is reversed.
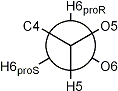
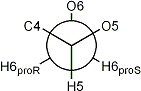
Newman projection of the hydroxymethyl rotamers in D-hexopyranoses
|
|
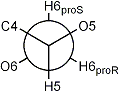
|
| gauche-trans rotamer |
gauche-gauche rotamer |
trans-gauche rotamer |
| 3JH5,H6proR large |
3JH5,H6proR small |
3JH5,H6proR small |
| 3JH5,H6proS small |
3JH5,H6proS small |
3JH5,H6proS large |
In galactose derivatives the chemical shift difference between H6proR and H6proS
is small which makes both
the assignment and the measurement of the coupling constants difficult.
Other methods that have been used with varying success include the optical rotation
and other coupling constants such as 3JCH.
In general they confirm the results of the analyses based on 3JHH
but are more prone to errors.
The results from the conformational analysis can be summarized as follows:
-
The most important interaction is that between the O6 oxygen and the substituent at C4 (in hexopyranoses,
C3 in pentofuranoses). 1,3-interactions between the terminal oxygen atom and ring substituents,
i.e. parallell arrangements of the C6-O6 and C4-O4
bonds, are avoided. Thus, gluco-derivatives have very little tg rotamer and
galacto-derivatives little gg-rotamer.
This is refered to as the Hassel-Ottar effect
(strictly this refers to 1,3 interactions in a six membered ring).
-
The gauche effect causes the population in the gg and gt rotamers,
which have O6 gauche to O5, to be favoured compared to tg.
The reason is the unfavourable interaction between the antibonding orbitals
of the C5-O5 and C6-O6 bonds in the trans-gauche rotamer.
-
The anomeric configuration has some influence on the rotamer distribution (and vice versa) but the
difference between α and β anomers approaches the accuracy of the conformational analyses.
The effect appears to be larger in furanosed than in pyranoses.
-
The rotamer distribution of the C6-O6 bond can be estimated from the
2JHH values. Small 2JHH
values suggest an anti arrangement of the C5-C6 and O6-X bonds. Most derivatives seem to have a
roughly equal amount of all three rotamers but the trans rotamer becomes favoured if the substituent
at O6 is very large, e.g. trityl.
There seems to be little, if any correlation with the ω angle.
The reason that galacto-derivatives have smaller 2JHH values than
gluco-derivatives is the small effect of C5-C6 rotamer distribution on the magnitude of
this coupling and not the presence of more trans rotamer.
|
|





